Allurion Expands Virtual Care Suite to the United States
Allurion Expands Virtual Care Suite to the United States
Virtual Care Suite (VCS) is now available to license in the United States for providers offering GLP-1 and other anti-obesity medication therapy, bariatric surgery, and weight loss devices
US product launch to kick off at the Obesity Medicine Conference in Denver on April 25
NATICK, Mass. – April 4th, 2024 – Allurion Technologies, Inc. (NYSE: ALUR), a company dedicated to ending obesity, today announced the commercial availability of its comprehensive Virtual Care Suite (VCS) in the United States.
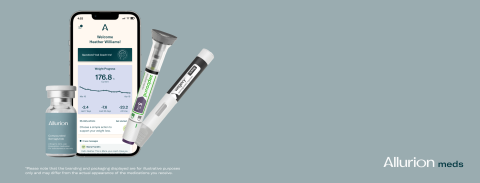
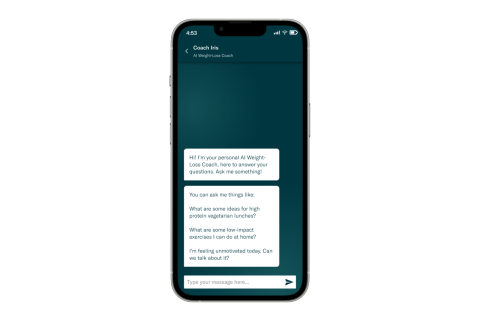
Already in use by thousands of providers in over 50 countries, the VCS is a dynamic weight-loss management suite powered by artificial intelligence (AI) featuring remote patient monitoring, telehealth and care team collaboration all under the one digital umbrella, designed to maximize patient outcomes and satisfaction. In 2023, Allurion added Coach Iris, a 24/7 AI-powered weight loss coach, to the VCS, enabling automated, conversational coaching and a more personalized behavior change program tailored to patient needs.
"After witnessing the profound impact our technology has had globally, we are thrilled to launch the VCS in the United States. Our team is incredibly excited to provide US clinics with digital solutions to help them significantly bolster both clinical outcomes and patient experience for all weight-loss treatments, including anti-obesity medications and bariatric surgery, especially as GLP-1 usage surges,” said Brian Conyer, Vice President of Digital Health. “Our research indicates that the VCS has already enhanced patient results, engagement, and satisfaction. Clinics with the highest VCS usage achieve an additional 11% weight loss1, with 16% more patients reaching their goals1, and a 17% increase in Net Promoter Score (NPS)2.”
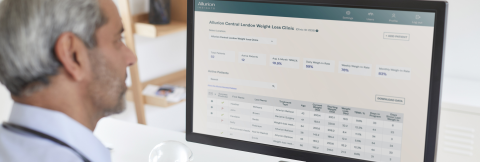
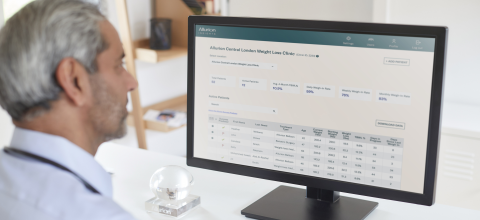
The VCS will be presented for the first time in the United States at the Obesity Medicine Conference in Denver on April 25, where providers will have the opportunity to request 1:1 meetings to discover the solution. As part of the launch, Allurion will also be unveiling a new version of Allurion Insights, the web-based dashboard that providers use to navigate the VCS.
The new version of Allurion Insights includes:
- Responsive, mobile app-like design: for providers and care teams on the move who want to stay in sync with their patients on their mobile phones
- More efficient navigation: thoughtfully organized by user activities, the updated navigation experience is designed to create more seamless access to a number of different data streams for individual patients
- Multitasking: for busy providers, multitasking allows them to document a clinical note or launch a video consultation while seamlessly viewing other patient information
“As use of anti-obesity medication in the United States increases, providers are being challenged to offer close follow-up and lifestyle modification coaching at scale,” said Shantanu Gaur, Founder and CEO. “We are thrilled to make the VCS available in the US and believe it could become an integral part of practices offering weight loss therapy of any kind.”
Sources: 1. Allurion Internal PEX Report, March 2023; 2. Allurion Internal Business Excellence Analysis, Q4 2023
About Allurion
Allurion is dedicated to ending obesity. The Allurion Program is a weight loss platform that features the Allurion Gastric Balloon, the world’s first and only swallowable, procedure-less intragastric balloon for weight loss, and offers access to the Allurion Virtual Care Suite, including the Allurion Mobile App for consumers, Allurion Insights for health care providers featuring the Coach Iris AI Platform, and the Allurion Connected Scale. The Allurion Virtual Care Suite is also available to providers separately from the Allurion Program to help customize, monitor and manage weight loss therapy for patients regardless of their treatment plan: gastric balloon, surgical, medical or nutritional. The Allurion Gastric Balloon is an investigational device in the United States.
For more information about Allurion and the Allurion Virtual Care Suite, please visit www.allurion.com
Allurion is a trademark of Allurion Technologies, Inc. in the United States and countries around the world.
Forward-Looking Statements
This press release may contain certain forward-looking statements within the meaning of the U.S. federal and state securities laws. These forward-looking statements generally are identified by the words “believe,” “project,” “expect,” “anticipate,” “estimate,” “intend,” “strategy,” “future,” “opportunity,” “plan,” “may,” “should,” “will,” “would,” “will be,” “will continue,” “will likely result,” and similar expressions and include statements regarding Allurion’s expectations for, and market acceptance of, Allurion’s Virtual Care Suite and the Allurion Program in general, Allurion’s ability to scale its business and in particular for the VCS, and the ability of the VCS to improve clinical and patient outcomes. Forward-looking statements are predictions, projections and other statements about future events that reflect the current beliefs and assumptions of Allurion’s management based on information currently available to them and, as a result, are subject to risks and uncertainties. Many factors could cause actual future results or developments to differ materially from the forward-looking statements in this communication, including but not limited to (i) the ability of Allurion to obtain and maintain regulatory approvals for and successfully commercialize the Allurion Program, including the Allurion Balloon and the VCS, (ii) the timing of, and results from, our clinical studies and trials, (iii) the evolution of the markets in which Allurion competes, (iv) the ability of Allurion to defend its intellectual property, (v) the impact of the COVID-19 pandemic, the Russia and Ukraine war, and the Israel-Hamas conflict on Allurion’s business, (vi) Allurion’s expectations regarding its market opportunities, including those for the VCS platform, and (vii) the risk of economic downturns and a changing regulatory landscape in the highly competitive industry in which Allurion operates. The foregoing list of factors is not exhaustive. You should carefully consider the foregoing factors and the other risks and uncertainties described in the “Risk Factors” section of Allurion’s Annual Report on Form 10-K filed on March 26, 2024 and other documents filed by Allurion from time to time with the U.S. Securities and Exchange Commission. These filings identify and address other important risks and uncertainties that could cause actual events and results to differ materially from those contained in the forward-looking statements. Forward-looking statements speak only as of the date they are made. Readers are cautioned not to put undue reliance on forward-looking statements, and Allurion assumes no obligation and does not intend to update or revise these forward-looking statements, whether as a result of new information, future events, or otherwise. Allurion does not give any assurance that it will achieve its expectations.
Contacts
Global Media
Cedric Damour
PR Manager
+33 7 84 21 02 20
cdamour@allurion.com
Investors
Mike Cavanaugh, Investor Relations
ICR Westwicke
(617) 877-9641
mike.cavanaugh@westwicke.com
Allurion Announces NYSE Acceptance of Plan to Regain Listing Compliance
NATICK, Mass. – December 2, 2024 – Allu...
Allurion Launches in the US with its First AI-Native Compounded GLP-1 Program
AllurionMeds is designed to...
Allurion Announces Submission of First Three Modules of Premarket Approval Application to the U.S. Food and...
Allurion Announces Publication of New Data Demonstrating Muscle Mass Preservation in Patients Experiencing 15%...
Allurion Announces Publication of Meta-Analysis Demonstrating the Safety and Effectiveness of the Allurion...
Allurion Announces Publication of New Data Demonstrating Weight Loss in Adolescents Suffering from Obesity
In a...
Allurion Announces Publication of New Data Demonstrating Continued Weight Loss at One Year On the Allurion...
Allurion Announces the Appointment of Eli Lilly Veteran, Keith Johns, to its Board of Directors
Mr. Johns has...
Allurion Announces Publication of New Data Demonstrating Lean Mass Gains in Patients Experiencing 14% Weight...
Allurion Announces Three Scientific Presentations at the 40th Annual Meeting of the American Society for...
Allurion Announces the Appointment of Ojas A. Buch as Chief Operating Officer
NATICK, Mass. -- June 5, 2024 -- ...
Allurion Announces Expansion of AI Weight Loss Coach to Support Patients on GLP-1s
Company also announces first...
Allurion Announces Three Scientific Presentations at the European Congress on Obesity, Including Study on Lean...
Allurion Announces Three Scientific Presentations at IFSO-European Chapter 2024, Including
First-Ever 3-Year...
Brendan Gibbons Appointed Chief Legal Officer at Allurion Technologies
NATICK, Mass. -- Jan. 29, 2024 -- Alluri...
Somerset NHS Foundation Trust Treats First Patients with Allurion Balloon for Pre-Surgical Weight Loss
Allurion...
Allurion's Virtual Care Suite Digital Platform Accelerates Commercial Expansion With New Strategic...
Allurion Program led to resolution of type 2 diabetes, hypertension, and obstructive sleep apnea in just 4...
New research highlights the positive impact of GLP-1s and other anti-obesity medications on the Allurion...
Allurion’s Digital Platform Gains Traction with Expanded Strategic Partnership
Company also announces addition...
Allurion Completes Enrollment in Pivotal AUDACITY Trial for the Allurion Balloon
AUDACITY trial to support FDA...
Allurion Announces Acceptance of Five Abstracts for Presentation at Obesity Week 2023
Research highlights...
Allurion Featured at the 2023 International Bariatric Club
Oxford University World Congress
Congress...
Allurion Introduces Coach Iris: A 24/7 Generative AI-Powered Weight Loss Coach
Powered by GPT and Fine-Tuned...
Allurion Announces Acceptance of Nine Scientific Presentations at IFSO 2023 World Congress
New data includes...
Allurion Debuts as a Publicly Traded Company on the NYSE
Allurion to commence trading on August 2, 2023 on the...
Allurion Announces New Additions to Board of Directors and Executive Leadership Team
Serial healthcare...
Allurion Presents New Data at the 2023 ASMBS Annual Meeting Demonstrating Mean Weight Reduction of Over 20%
P...
Allurion Announces Collaboration Agreement with Medtronic to Expand Access to AI-Powered Weight Loss Program
...Allurion to Participate in the Jefferies Healthcare Conference
NATICK, Mass. -- May 30, 2023 – Allurion, a...
First Multi-Center Study Combining the Allurion Program with GLP-1 Therapy Presented at the European Congress...
Allurion Presents Findings from Landmark Study Evaluating Fully Remote Weight Loss Coaching Program
Proprietar...
Allurion Announces Premium Tiers for the Virtual Care Suite and Launch of Proprietary “My Daily Action” Bundle...
Allurion Announces Appointment of MIT Distinguished Professor for AI and Health and MacArthur “Genius” Fellow
...Allurion named “Champion Partner” of the World Obesity Federation and official sponsor of World Obesity Day...
Allurion, a Global Leader in Weight Loss Technology, to Become Publicly Listed Through Business Combination...
Allurion Technologies’ training awarded World Obesity Federation SCOPE accreditation
- Allurion’s gold...
Company reports 444% revenue growth from 2018 to 2021 and has also been named as one of MedTech Outlook’s Top...
- The world’s first and only procedureless gastric balloon receives approval from Brazilian Health...
New Delhi, 06 Sep 2022:
Allurion, a company dedicated to ending obesity, has launched the Allurion...
- Company launches the Allurion Iris™ Artificial Intelligence Platform and introduces Success Predictor, a...
Company announces approval of the Allurion Balloon in Canada, Mexico, Australia, and India where approximately...